
What if you could isolate a single cancer cell from a patient and use its genetic makeup to create a personalized treatment plan optimized for that individual?
That’s exactly what Menarini Silicon Biosystems Inc. (MSB) hopes its technology could one day do for people with multiple myeloma.
Multiple myeloma is the most common hematological malignancy. It forms in plasma cells, white blood cells found mainly in the bone marrow that protect the body from infection by producing antibodies. When these cells become malignant, abnormal plasma cells accumulate in the bone marrow, producing abnormal antibodies and crowding out normal blood-forming cells. Some of these abnormal cells, known as circulating multiple myeloma cells (CMMC), escape from the primary tumor space and travel through the bloodstream.
Current approaches for diagnosing patients with multiple myeloma require bone marrow aspiration and biopsy. These are invasive procedures that use a hollow needle inserted into the hipbone or breastbone to remove bone marrow, blood, and a small piece of bone. While generally safe, bone marrow exams can result in excessive bleeding, infection, and long-lasting discomfort.
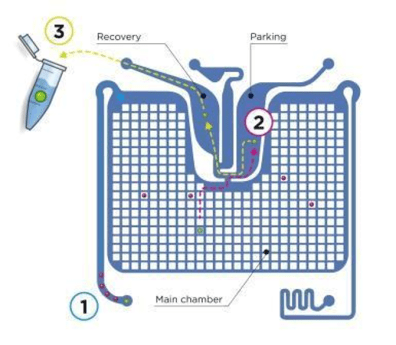
Liquid biopsies, on the other hand, examine cancer-related material from a peripheral blood sample obtained from a simple, common blood draw. MSB has developed a method for enumerating CMMC from liquid biopsies using CELLSEARCH® and DEPArray™ technologies.
CELLSEARCH is the first and only clinically validated, FDA-cleared blood test for enumerating circulating tumor cells.* The CELLSEARCH platform can separate CMMC from a blood sample, producing an enriched population of CMMC to be analyzed with DEPArray technology, an automated image-based cell selection platform that can recover single cells of interest from heterogeneous samples for downstream molecular analysis.
In 2018, a study published in the British Journal of Hematology found that the assay could be used to monitor progression, especially in early stage asymptomatic disease, and minimal residual disease. Researchers counted CMMCs from more than 1,000 patient samples, including both newly diagnosed multiple myeloma and high/intermediate risk smoldering multiple myeloma with clinical follow‐up data.
Enumerating CMMC in patients with or suspected of having multiple myeloma could allow a physician to stage the disease and monitor progression in a noninvasive way. This could enable them to capture those cells more frequently, potentially making it possible to monitor the disease in real time.
Molecular profiling of the recovered CMMC may help determine specific genetic abnormalities in a particular patient, potentially allowing for personalized treatment. Molecular analysis of CMMC could also be used after treatment to detect remaining, rare tumor cells to see how the disease has modified itself and if a patient may benefit from an additional treatment course.
The combination of CELLSEARCH and DEPArray technologies to recover and analyze CMMC could one day allow for a robust liquid biopsy analysis of multiple myeloma for detection of disease, treatment planning, and real-time monitoring in a noninvasive manner.
The CMMC assay is currently available as a laboratory service at MSB. As experts in rare cell and assay development, MSB provides global clinical laboratory services using its CLIA-certified and ISO-accredited lab. These oncology focused services are tailored to meet unique client needs by offering In Vitro Diagnostic, Research Use Only, and Lab Developed Test services. The group performs and develops a diverse range of assays for pharma and academia partners worldwide utilizing both CELLSEARCH and DEPArray technologies. MSB’s expert team has over 19 years pharma services development, with clinical trial testing experience used to develop assays that fit exact study needs.
MSB will be releasing a commercially available Research Use Only CMMC Kit using the current CellSearch platform in the summer of 2020.
*CELLSEARCH is currently FDA approved for use in patients with metastatic breast, prostate, and colorectal cancer. CELLSEARCH technology for detection of CMMC is for research use only.